Carbon Nanotubes (CNTs): Properties, Importance, Manufacturing, Challenges, and Real-World Applications Explained
By Mufaddal Shakir
3/5/202612 min read
Introduction
Carbon nanotubes (CNTs) are increasingly gaining popularity and are currently one of the most talked about materials in the field of nanotechnology due to many good reasons. CNTs combine ultra-low weight with extraordinary strength, have high electrical conductivity, and have excellent thermal properties. Owing to this rare property package, CNTs are actively commercialized and used in the areas ranging from conductive composites and lithium-ion batteries to sensors, EMI shielding, and next-generation electronics.
In this article, you will learn everything about CNTs, from their discovery, significance, properties, and manufacturing to the challenges associated with them and their real-world applications.
1. What are Carbon Nanotubes (CNTs)?
Carbon nanotubes are cylindrical nanostructures made up of carbon atoms that are arranged in a hexagonal lattice (similar to graphene) and “rolled” into a tube. Their diameter is typically on the nanometer scale, while their length can reach micrometers to millimeters, giving CNTs an extremely high aspect ratio (length/diameter). This geometry is a main reason they form percolating networks in composites and conductive coatings.
Types of Carbon Nanotubes:
a) Single-Walled Carbon Nanotubes (SWCNTs): They consist of a single graphene sheet that is rolled into a cylinder with a diameter ranging from 1 to 2 nm. SWCNTs are known for their high electrical conductivity and their high tensile strength. These are usually quite expensive to produce.
b) Multi-Walled Carbon Nanotubes (MWCNTs): They consist of multiple concentric graphene cylinders, which have diameters in the range of 2-100 nm. MWCNTs are generally stiffer and are easier to produce at scale and cost-effective in comparison to SWCNTs. They are widely utilized as structural reinforcements in composites and are used for applications that require high strength and stiffness.
Both types of CNTs possess exceptional mechanical, thermal, and electrical properties. CNTs have the tendency to behave as metallic or semiconducting depending on their atomic arrangement (chirality), which is important for nanoelectronics and sensors.


Single-walled and Multi-walled carbon nanotubes (CNTs)
2. How were CNTs discovered?
Although carbon-related nanostructures were discussed earlier in different forms, CNTs gained global scientific attention after Sumio Iijima, a Japanese physicist and inventor, reported needle-like graphitic microtubules in 1991, which led to the discovery of carbon nanotubes. The tubes were noticed in carbon soot produced by an arc-discharge evaporation method (utilizing graphite electrodes) in a helium atmosphere, a method originally used to create fullerenes. Sumio made use of the Transmission Electron Microscope (TEM) to observe the soot, noticing the hollow and needle-like microscopic structures. Initial observations led to the discovery of MWCNTs, with SWCNTs being discovered shortly after in 1993.
The discovery of CNTs in 1991 highlighted the unique atomic structure, which led to intensive research into their high mechanical and electrical properties. Later reviews and historical analyses helped consolidate CNTs as a foundational material class in modern nanoscience and nanotechnology.


An arc discharge method for producing CNTs
3. Why are CNTs so Important?
Carbon nanotubes are of importance owing to their exceptional mechanical, electrical, and thermal properties, which position them as "advanced materials" for next-generation technology. CNTs matter because they provide an unusually powerful combination of:
High-aspect-ratio structure
Excellent reinforcement properties
High thermal conductivity
High electrical conductivity
Large surface area
Chemical tunability
These unique characteristics make CNTs an ideal choice for “multifunctional materials.” For instance, a polymer composite that is not only mechanically strong but also electrically conductive, EMI-shielding, and self-sensing (damage detection via resistance changes). Modern reviews highlight the importance of CNTs as both structural reinforcements and functional fillers, especially in composite materials.
4. What are the Key Properties of CNTs?
a) Mechanical Properties
CNTs are famous for their high strength and stiffness relative to their weight. In practice, the mechanical performance you get in a real composite depends heavily on dispersion quality, alignment, interfacial bonding, and CNT defects—so engineering matters as much as the nanotubes themselves. Many studies report the theoretical tensile strength in the range of 50-150 GPa and Young's modulus value in the range of 0.9-1 TPa. However, the measured tensile properties of CNTs are much lower. A study published in the journal Scientific Reports concluded the average tensile strength and Young's modulus value of MWCNTs to be 0.85 GPa and 34.65 GPa respectively, showing a huge contrast between the theoretical and experimental values.
b) Electrical Properties
The electrical properties of CNTs heavily depend on their chirality. A few SWCNTs are metallic, that is, they have zero bandgap, while others are semiconducting, having a finite bandgap. The reported measured conductivity value for metallic SWCNTs is often on the order of 10⁵–10⁷ S·m⁻¹ in optimized conditions. High-performance CNT fibers and films can reach up to 10⁵–10⁷ S·m⁻¹ (Scientific Reports).
c) Thermal Properties
Heat in CNTs is mainly transported by phonons (lattice vibrations). Intrinsic axial thermal conductivity of both SWCNTs and MWCNTs is reported in the range of 1000 to 2500-3000 W·m⁻¹·K⁻¹. The coefficient of thermal expansion (CTE) for CNTs is usually small, with reported values around ~1.9×10⁻⁵ to 2.1×10⁻⁵ K⁻¹ (ScienceDirect).
d) Chemical Properties
CNTs are chemically similar to graphene/graphite. They are also known to be chemically inert in many environments. Oxidation or high temperatures introduce defects that alter their mechanical, electrical, and thermal properties. CNTs mostly begin to oxidize/lose mass in air at elevated temperatures around ~500 ℃. Much research has observed oxidation starting at around ~450 ℃ with full combustion occurring at high temperatures (MDPI).
e) Surface Properties
CNTs have a large surface area and can be chemically modified (functionalized) to improve bonding with polymers and compatibility with solvents. Studies report the specific surface area for SWCNTs to be around ~150 to 600 m²·g⁻¹ and ~15 to 300 m²·g⁻¹ for MWCNTs. Pure CNT mats are known to be hydrophobic. However, functionalized CNTs can display water contact angles from ~40° (hydrophilic) to >140° (hydrophobic) depending upon the chemistry.
5. Synthesis of CNTs
CNTs are generally synthesized utilizing high-temperature processes that break down carbon-containing solids or gases and allow carbon atoms to assemble into cylindrical graphene structures. CNTs are synthesized using many physical and chemical methods that regulate their structure (SWCNTs or MWCNTs), purity, and yield. The three main techniques, such as Chemical Vapor Deposition (CVD), Arc Discharge, and Laser Ablation remain the most utilized in research and industry. These methods are described in detail below:
a) Arc Discharge Technique:
This was the earliest synthesis technique, which was developed in 1991. In this technique, an electric arc is struck between two high-purity graphite electrodes (cathode and anode) in an inert gas atmosphere such as argon or helium. The high-temperature plasma (up to 6000 K) vaporizes the anode's carbon, which then condenses on the cooler cathode to form CNTs. Without a catalyst, this method primarily yields MWCNTs, whereas, with metal catalysts, SWCNTs are produced.
Advantages:
Structural defects are less
Enhanced mechanical and electrical properties
High crystalline CNTs are produced
Disadvantages:
Soot and impurities are produced
Purification is required
Energy intensive and costly
b) Chemical Vapor Deposition (CVD):
This is the most dominant commercial CNT manufacturing method today. A hydrocarbon gas (such as methane, ethylene, or acetylene) is decomposed over metal catalyst nanoparticles (Fe, Ni, or Co) at temperatures ranging between 600°C and 1200°C. Carbon atoms from the decomposed gas diffuse into or over the catalyst particles and precipitate to form nanotubes. Depending on the interaction between the catalyst and the substrate, growth follows a tip-growth (catalyst lifts as CNT grows) or base-growth (catalyst remains attached to substrate) model.
Advantages:
Great for mass production
Low cost and scalable
Good control over length and alignment
Disadvantages:
Requires the removal of catalyst
Purity of CNTs depends on process control
More structural defects than arc discharge and laser ablation method
C) Laser Ablation Technique:
This method is known for creating high-purity SWCNTs with controllable diameters. A high-power pulsed or continuous-wave laser (such as Nd:YAG) vaporizes a graphite target doped with metal catalysts in a high-temperature furnace (approximately 1200°C). An inert carrier gas sweeps the vaporized carbon to a water-cooled collector where it condenses into nanotubes.
Advantages:
High-purity SWCNTs are produced
Very few defects are developed
High structural quality
Disadvantages:
Consumes high energy
Not suitable for mass production
Equipment is expensive
Following are some of the latest and advanced techniques:
d) HiPco (High-Pressure Carbon Monoxide):
Developed at Rice University, HiPco is a gas-phase CVD method specifically optimized for producing high-purity SWCNTs. A high-pressure carbon monoxide (CO) gas (typically 30–50 atm) is mixed with a small amount of iron pentacarbonyl in a high-temperature reactor (900–1100°C). The iron precursor decomposes in the gas phase to form "floating" iron clusters that act as catalytic seeds. CNTs then grow via the Boudouard reaction (2CO -> C + CO2) on these clusters. It produces very small diameter SWCNTs (approximately 1 nm) with high structural quality and minimal amorphous carbon.
e) Floating Catalyst CVD (FCCVD):
FCCVD is a continuous, substrate-free technique where both the catalyst and the carbon source are introduced as vapors or aerosols into the reactor. A metallocene (such as ferrocene) and a hydrocarbon (such as methane or ethylene) are injected into a high-temperature furnace (approximately 1200°C). Catalyst nanoparticles form in situ within the gas flow rather than on a fixed surface. CNTs grow as they "float" through the reaction zone, eventually forming an aerogel-like "sock" or "smoke." It is the primary method for allowing the continuous production of macroscopic CNT materials such as films, ropes, and sheets.
f) Plasma-Enhanced CVD (PECVD):
PECVD uses an electric field to create a plasma (ionized gas), which provides additional energy to the system, allowing for growth at lower temperatures. A plasma is generated in a gas mixture (e.g., methane and ammonia) over a substrate pre-coated with catalyst nanoparticles. The plasma dissociates the precursor gas molecules into highly reactive radicals. The electric field within the plasma sheath creates a strong aligning force. It enables the growth of Vertically Aligned Carbon Nanotubes (VACNTs), where the tubes grow perpendicular to the substrate like a "forest." This is important for applications such as field emission displays and interconnects in electronics.
g) Emerging Techniques:
Liquid Electrolysis: This technique utilizes molten carbonates and electric current to convert into high-value CNTs, serving as a carbon capture technology.
Flame Synthesis: This method develops CNTs in controlled combustion environments (for example, burning methane or ethylene), providing a potential path for low-cost and large-scale synthesis.
Bio-precursor Synthesis: In this process, renewable resources such as camphor oil, castor oil, or even plant waste (biochar) are used as sustainable carbon sources for CVD.
Ball Milling: A mechanical method where graphite powder is crushed into nucleation structures and then annealed to form nanotubes.
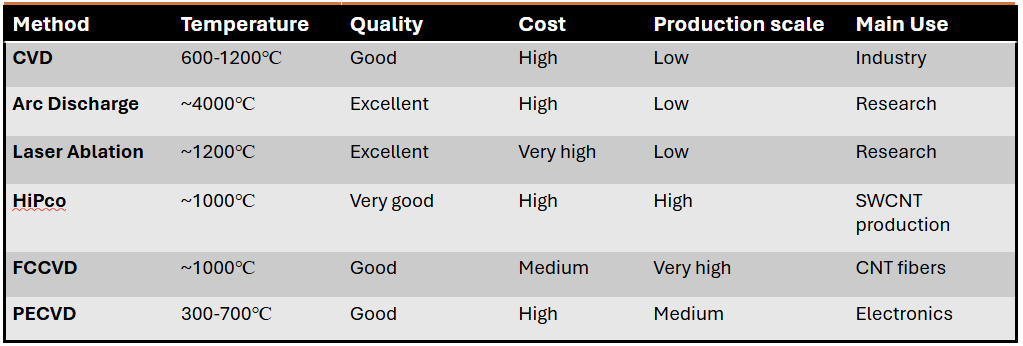
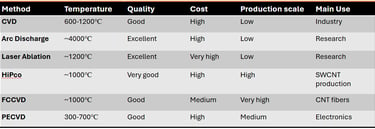
Comparison of different synthesis methods of CNTs
6. Real-World Applications of CNTs
CNTs are used across different industrial sectors such as biomedical, electronics, automobiles, aviation, and energy for their superior mechanical, thermal, and electrical properties. Some real-world applications of CNTs in various industries are discussed below:
a) Automotive Industry:
CNT-reinforced polymer composites are utilized in automotive fuel systems to avoid dangerous electrostatic discharge and sparks. A few examples of parts include fuel connectors, fuel tank liners, and polymer fuel lines.
CNT additives are used in automotive primers and coating paint systems, which improves the conductivity and coating quality.
CNTs are used as conductive additives in lithium-ion batteries, dramatically increasing production demand with components including battery anode and cathode.
b) Aviation Industry:
CNT polymer composites are used in structural components, EMI shielding, and thermal management systems. CNTs improve conductivity without adding weight and reduce fuel consumption.
The Boeing Company has patented the use of CNTs for structural health monitoring of composite materials utilized in aircraft structures (US9329021B1).
c) Energy Industry:
CNTs are commercially utilized in battery manufacturing, as they improve the charge rate, cycle life, and capacity of lithium-ion batteries.
CNTs are also utilized in supercapacitors and fuel cells as materials in electrode films, current collectors, and catalyst supports.
Wind turbine blades are coated with CNT coatings to improve structural durability from corrosion resistance and as conductive coatings to protect from the lightning strike.
d) Electronics Industry:
IMEC is using carbon nanotubes for Pellicles (an ultra-thin, ultra-lightweight, semi-transparent mirror) in semiconductor lithography.
Wearable electronics and 5G/6G communication. Electrodes with SWCNTs exhibit excellent electrochemical properties and flexibility.
Utilization of carbon nanotubes as the channel material of carbon nanotube field-effect transistors.
e) Biomedical Industry:
In tissue engineering, carbon nanotubes are utilized as scaffolds for bone growth. CNTs provide high strength, and also their nanoscale structure mimics collagen fibers.
CNTs have been shown to increase the effectiveness of bioactive coatings for the attachment, proliferation, and differentiation of Osteoblasts, and have been used as a bone substitution material.
Utilization of SWCNTs as highly sensitive nanosensors for a variety of important biomolecules. Examples include detection of reactive nitrogen and oxygen species and other small molecules, proteins, lipids, enzymes, as well as bacteria.
f) Other applications:
CNT-modified asphalt road pavement was built, and the real road test showed a 67% increase in crack resistance and ruts, thereby increasing the lifespan of the materials.
A CNT composite vessel, called the Piranha Unmanned Surface Vessel, was also developed by Zyvex Technologies. The weight of the vessel was 8,000 lb, having a payload capacity of 15,000 lb over a range of 2,500 miles, demonstrating the real structural use of CNTs in transportation.


The pellicle mirror of the Canon EOS RT (Wikipedia)
7. Challenges and Limitations of CNTs
There are significant challenges faced by CNTs, typically in terms of high production costs, difficulty in purification, and achieving uniform dispersion within composite materials. Some of the major challenges involved with CNTs are addressed below:
Dispersion and Agglomeration: CNTs like to bundle together due to strong van der Waals forces, which makes them difficult to disperse uniformly, ultimately affecting their mechanical, thermal, and electrical properties.
Interfacial bonding: Owing to their nature of chemical inertness, it is difficult for CNTs to form strong bonds with metals or polymer matrices. This poor interfacial bonding can reduce the performance of the material.
Chirality control: It is difficult to control the specific chirality (such as diameter and angle) of SWCNTs, which is an important criterion in selecting between metallic and semiconducting behaviour.
Toxicity and Health Risks: CNTs possess huge health risks, typically when inhaled, as they can cause inflammation similar to asbestos fibers. Their large surface area also indicates that they can strongly interact with cells.
Purification and Production: It is tough to produce defect-free and high-quality CNTs at large industrial scales, as CNTs generally contain impurities (metal catalysts such as Ni, Fe, and Co), and amorphous carbon, requiring expensive and complex production routes.
Conclusion
Carbon nanotubes have earned their reputation and emerged as a transformational nanomaterial because they combine lightweight strength, electrical conductivity, thermal performance, and chemical flexibility in a single structure. From CNT composites in transportation to CNTs in lithium-ion batteries, sensors, and EMI shielding, their impact is already real and still growing.
At the same time, the high performance of CNTs is not magical. Different factors such as dispersion, processing, and interface engineering determine whether you get a true advantage of their properties in real-world applications. Also, unlike any other nanomaterials, responsible handling and safety practices do matter. With improved manufacturing control and smarter composite design, CNTs will continue to move from labs into scalable, high-value applications across energy, electronics, and advanced composite materials.
FAQs
1. Why are CNTs important for battery applications?
Answer: CNTs enhance mechanical stability, electrical conductivity, and charge transport in lithium-ion battery electrodes. They help form conductive networks that improve fast charging, power delivery, and cycle life.
2. Are CNTs considered stronger than steel?
Answer: CNTs are significantly stronger than steel in terms of weight. CNTs have exceptionally high tensile strength and stiffness, placing them as a perfect candidate for reinforcement in lightweight composites, though their performance in the real world depends on dispersion and bonding.
3. Are CNTs better in terms of safety?
Answer: The safety of CNTs usually depends on the exposure route, particle form, and concentration. Although CNTs are safe when incorporated in solid materials (such as composites), inhalation of airborne CNT particles during manufacturing must be controlled using proper ventilation and safety protocols.
4. What is the cost of CNTs?
Answer: CNTs are extremely expensive, with the price of industrial-grade MWCNTs approximately ranging from $50 to $500/kg and high-purity SWCNTs ranging from $1000 to $5000/kg. The prices highly depend on the type, purity, and quantity.
References:
1. Dresselhaus, M. S., Dresselhaus, G., Eklund, P. C., & Rao, A. M. (2000). Carbon nanotubes. In The physics of fullerene-based and fullerene-related materials (pp. 331-379). Dordrecht: Springer Netherlands. https://doi.org/10.1007/978-94-011-4038-6_9
2. Popov, V. N. (2004). Carbon nanotubes: properties and application. Materials Science and Engineering: R: Reports, 43(3), 61-102. https://doi.org/10.1016/j.mser.2003.10.001
3. Ajayan, P. M., & Zhou, O. Z. (2001). Applications of carbon nanotubes. Carbon nanotubes: synthesis, structure, properties, and applications, 391-425. https://doi.org/10.1007/3-540-39947-X_14
4. De Volder, M. F., Tawfick, S. H., Baughman, R. H., & Hart, A. J. (2013). Carbon nanotubes: present and future commercial applications. Science, 339(6119), 535-539. https://doi.org/10.1126/science.1222453
5. Kostoglou, N., Tampaxis, C., Charalambopoulou, G., Constantinides, G., Ryzhkov, V., Doumanidis, C., ... & Rebholz, C. (2020). Boron nitride nanotubes versus carbon nanotubes: A thermal stability and oxidation behavior study. Nanomaterials, 10(12), 2435. https://doi.org/10.3390/nano10122435
6. Birch, M. E., Ruda-Eberenz, T. A., Chai, M., Andrews, R., & Hatfield, R. L. (2013). Properties that influence the specific surface areas of carbon nanotubes and nanofibers. Annals of occupational hygiene, 57(9), 1148-1166. https://doi.org/10.1093/annhyg/met042
7. Nagaya, H., Cho, J., & Hori, T. (2021). Thermal conductivity of single-walled carbon nanotubes under torsional deformation. Journal of Applied Physics, 130(21). https://doi.org/10.1063/5.0073709
8. Yahyazadeh, A., Nanda, S., & Dalai, A. K. (2024). Carbon nanotubes: a review of synthesis methods and applications. Reactions, 5(3), 429-451. https://doi.org/10.3390/reactions5030022
9. Sajid, M., Asif, M., Baig, N., Kabeer, M., Ihsanullah, I., & Mohammad, A. W. (2022). Carbon nanotubes-based adsorbents: Properties, functionalization, interaction mechanisms, and applications in water purification. Journal of Water Process Engineering, 47, 102815. https://doi.org/10.1016/j.jwpe.2022.102815
10. Kolahdouz, M., Xu, B., Nasiri, A. F., Fathollahzadeh, M., Manian, M., Aghababa, H., ... & Radamson, H. H. (2022). Carbon-related materials: graphene and carbon nanotubes in semiconductor applications and design. Micromachines, 13(8), 1257. https://doi.org/10.3390/mi13081257
Connect
Discover
Email: automaterials53@gmail.com
© 2026. AutoMaterialsInsights. All rights reserved.
